

Ashwini Hospital Balewadi, Pune, is a leading healthcare facility known for its excellence in medical services. We provide state-of-the-art infrastructure, advanced medical equipment, and a team of skilled doctors, nurses, and healthcare professionals. Our hospital is dedicated to offering personalized and compassionate care, ensuring that every patient receives the best treatment in a comfortable and supportive environment. With a strong focus on innovation, patient satisfaction, and affordability, we strive to be the preferred healthcare provider in the region.
Happy Smiles
OurStaff
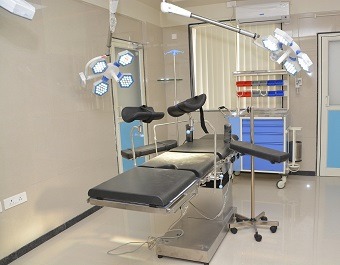
ClinicRooms
PresentMachines
The Obstetrics & Gynaecology (OB-GYN) Department at Ashwini Hospital is dedicated to providing comprehensive, compassionate, and specialized care for women at every stage of life.
Read MoreThe Fertility Department at Ashwini Hospital is a state-of-the-art facility equipped with the latest technology to provide accurate, reliable, and timely results
Read MoreThe Pediatrics Department at Ashwini Hospital is committed to providing comprehensive, compassionate, and child-friendly healthcare for newborns, infants, children, and adolescents.
Read MoreHolistic Care for Your Circle of Life


Ashwini hospital is located in Balewadi Pune. It is a multispeciality hospital with modern medical equipment. 24*7 Emergency Services. Multispeciality Hospital.
Multispeciality clinics are an extension of Ashwini Hospital located in Kothrud Pune.
“After taking treatment over 5 years in UK, I lost all the hope. But when I met Dr. Mithil Patil & followed his treatment over a few months, I got pregnant with very minimal assistance.”
“After visiting many more infertility centres, I decided to opt for adoption. Given a last try at De Novo Centre, in the first cycle of IUI I conceived!!”
“State-of-the-art IVF unit with all the facilities under one roof, making the dreams come true.”

Success Surgery


MD Obstetrics & Gynaecology FMF,UK Certified Sonologist ID : 72204
Dr. Mithil Patil
is a qualified & an experienced Gynecologist who has pursued his career in Infertility & Fetal Medicine. Passed out from Government Medical College , Solapur as a MD he has developed fine skills & superior clinical acumen . While working at Civil Hospital Solapur , he has performed & assisted many complicated surgeries & which has made him competent surgeon.
Quest for excellence has compelled him to undergo rigorous training in endoscopy at Cochin with Dr.Hafeez Rehman , renowed laparoscopic surgeon down south. He has further achieved milestone by doing fellowship training in Reproductive Medicine at Chennai Fertility Centre , Chennai with Dr.Thomas ( embryologist ) which has refined his skills in ART. Impressed by his work in Fetal Medicine, he has been certified by Fetal Medicine Foundation,UK as a certified sonologist( ID : 72204) for the prenatal screening method. He has been constantly working on the Infertility & fetal Medicine, bringing innovation in the varied methods & making treatment feasible to all class of the patients. He is currently working on a project named First Trimester Screening with Dr.S.Suresh , Chennai at Bharati Hospital & Research centre.
MD - General Medicine, MBBS
Dr. Ashwini Patil is a highly accomplished physician with a specialization in internal medicine. She holds a Doctor of Medicine (MD) degree in Medicine and has several years of experience providing quality healthcare to patients. Dr. Patil is known for her compassionate bedside manner and her dedication to delivering personalized care to each of her patients.
In her practice, Dr. Patil focuses on diagnosing and managing a wide range of medical conditions, from common ailments to complex diseases. She is skilled in conducting thorough evaluations, ordering diagnostic tests, and creating treatment plans tailored to each patient's specific needs. Dr. Patil is committed to staying abreast of the latest developments in medicine and continuously seeks to enhance her knowledge and skills through ongoing education and training.
Patients appreciate Dr. Patil for her attentive listening skills, empathy, and genuine concern for their well-being. She takes the time to educate her patients about their health conditions and works collaboratively with them to develop strategies for improving their overall health and wellness.
Outside of her clinical practice, Dr. Patil is involved in medical research and has published numerous articles in reputable medical journals. She is also actively involved in teaching and mentoring medical students and residents, passing on her knowledge and expertise to the next generation of healthcare professionals.
In summary, Dr. Ashwini Patil is a dedicated and skilled physician who is deeply committed to providing the highest standard of care to her patients. Her passion for medicine, combined with her professionalism and compassion, make her a trusted and respected healthcare provider in the community.


Ashwini Hospital is dedicated to providing comprehensive, high-quality, and patient-centric healthcare with a commitment to excellence. With a team of highly experienced doctors, skilled medical professionals, and state-of-the-art infrastructure, we ensure world-class treatment across multiple specialties. Our hospital is equipped with advanced diagnostic and surgical technologies, modern ICUs, and 24/7 emergency care, ensuring prompt and effective medical attention when you need it most. We prioritize personalized treatment plans, focusing on each patient’s unique needs while maintaining affordable and transparent pricing. At Ashwini Hospital, we blend cutting-edge medical advancements with compassionate care, making us a trusted healthcare destination for individuals and families. Whether it's routine check-ups, complex surgeries, or critical care, we are committed to your health and well-being every step of the way.
Our hospital is open 24/7 for emergencies. Outpatient services are available from 9 AM to 10 PM (Monday to Saturday).
We accept cash, credit/debit cards, and digital payment methods.
You can book an appointment online through our website or call us at 9975035364 . Walk-ins are also welcome.